Protect your family from respiratory illnesses. Schedule your immunization here >
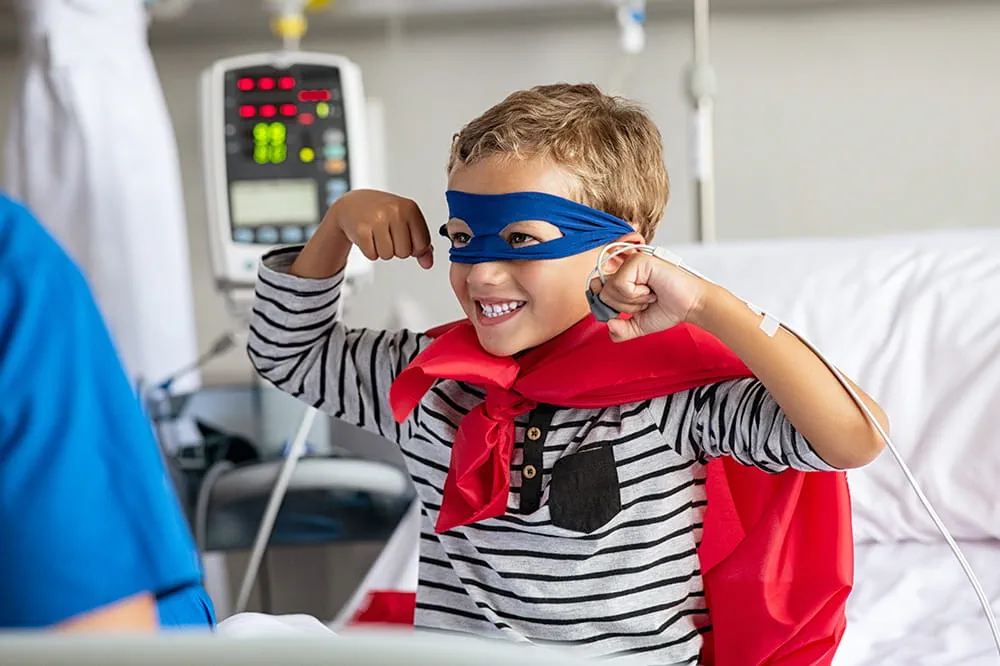
Ranked nationally in pediatric care.
Arkansas Children's provides right-sized care for your child. U.S. News & World Report has ranked Arkansas Children's in seven specialties for 2025-2026.

It's easier than ever to sign up for MyChart.
Sign up online to quickly and easily manage your child's medical information and connect with us whenever you need.
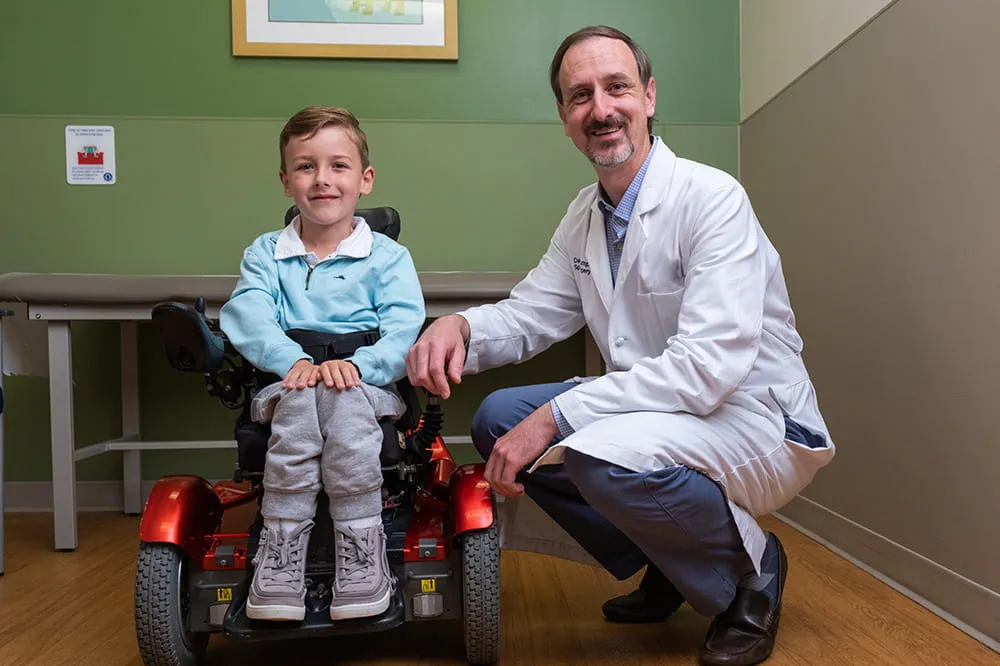
We're focused on improving child health through exceptional patient care, groundbreaking research, continuing education, and outreach and prevention.

When it comes to your child, every emergency is a big deal.
Our ERs are staffed 24/7 with doctors, nurses and staff who know kids best – all trained to deliver right-sized care for your child in a safe environment.

Arkansas Children's provides right-sized care for your child. U.S. News & World Report has ranked Arkansas Children's in seven specialties for 2025-2026.

Looking for resources for your family?
Find health tips, patient stories, and news you can use to champion children.

Support from the comfort of your home.
Our flu resources and education information help parents and families provide effective care at home.

Children are at the center of everything we do.
We are dedicated to caring for children, allowing us to uniquely shape the landscape of pediatric care in Arkansas.

Transforming discovery to care.
Our researchers are driven by their limitless curiosity to discover new and better ways to make these children better today and healthier tomorrow.

We're focused on improving child health through exceptional patient care, groundbreaking research, continuing education, and outreach and prevention.
Then we're looking for you! Work at a place where you can change lives...including your own.

When you give to Arkansas Children's, you help deliver on our promise of a better today and a healthier tomorrow for the children of Arkansas and beyond

Become a volunteer at Arkansas Children's.
The gift of time is one of the most precious gifts you can give. You can make a difference in the life of a sick child.

Join our Grassroots Organization
Support and participate in this advocacy effort on behalf of Arkansas’ youth and our organization.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.

Learn How We Transform Discovery to Care
Scientific discoveries lead us to new and better ways to care for children.
When you give to Arkansas Children’s, you help deliver on our promise of a better today and a healthier tomorrow for the children of Arkansas and beyond.

Your volunteer efforts are very important to Arkansas Children's. Consider additional ways to help our patients and families.

Join one of our volunteer groups.
There are many ways to get involved to champion children statewide.

Make a positive impact on children through philanthropy.
The generosity of our supporters allows Arkansas Children's to deliver on our promise of making children better today and a healthier tomorrow.

Read and watch heart-warming, inspirational stories from the patients of Arkansas Children’s.

Hello.

Arkansas Children's Hospital
General Information 501-364-1100
Arkansas Children's Northwest
General Information 479-725-6800

Resources for Researchers
Our Services
A service-oriented philosophy was in mind when the administrative staff was reorganized in 1997 in order to meet the dynamic needs of ACRI’s research community. The staff acts as facilitators to ease the administrative burden of sponsored research. At the same time, the office provides an array of unique services, outside the general area of research administration, that have proven to be quite beneficial to the Institute’s cause.
-
Research
Contact Our Administrative Staff
Bookmark Contact Our Administrative StaffQuestions about Arkansas Children's Research Institute? Our administrative team can help. Contact us today.
-
Research
Printable Research Guidebook
Bookmark Printable Research GuidebookDownload the Research Guidebook for detailed descriptions of research support services offered by ACRI Research Administration.
-
Research
ACRI 3-Year Roadmap
Bookmark ACRI 3-Year RoadmapThis addendum to the Arkansas Children’s Strategic Plan 2020-2025 lays out three thematically oriented goals, each employing the core strategies of the organization’s strategic plan to achieve measurable impact and transform our research enterprise.
Animal Forms:
For questions related to animal forms, please email harrisonblaket@uams.edu.
ACRI Grant Submission Forms:
For questions related to animal forms, please email YountPA@archildrens.org.
For the extramural budget template, please email our Grant Development Specialist.
Intramural Grant Application Forms:
For questions related to intramural grant application forms, please email McMainsAM@archildrens.org.
HIPAA Forms:
For questions related to HIPAA, please contact Amanda Holloway, CHRC , Human Research Protections Program Director by phone at 501-364-2862 or by email at hollowayag@archildrens.org.
Descriptions of Facilities Boilerplate Text
Descriptions of the facilities and grant resources are available to cut and paste into grant applications. If you would like a copy of these descriptions, please send an email to Phaedra or John and they will send those to you via email.The Biostatistics Core at ACRI provides comprehensive assistance with study design, data analysis, and statistical results interpretation. The core is staffed by two doctoral level statisticians and two master’s level statisticians who are available to support researchers of Arkansas Children’s with statistical applications related to basic science, clinical, health outcomes, epidemiologic, and behavioral research. Our statisticians have knowledge and expertise in SAS, open-source R, and SPSS software packages.
Key Services
- Assist with grant proposal development
- Determine study or experimental design
- Calculate sample size and power
- Identify appropriate statistical methods or models
- Analyze data and interpret results
- Develop surveys and questionnaires
- Review manuscripts for statistical content
- Assist in writing statistical content for grant proposals and publications
- Develop tables and figures for scientific publications
Fees
The first hour of consulting is free. Additional support will be charged on an hourly basis
Consulting for grant proposal preparation is free. Costs associated with statistical support needed during the project period should be included in the grant application budget.
- Intramural Applications: Proposal budgets should include a single charge for statistical support that is based on the approximate total hours of engagement.
- Extramural Applications: Proposal budgets should include salary support at the level of effort required to complete the work. Effort determinations will be based on the extent and volume of work (scope of the project). Work is defined as study design, drafting of data analysis plan, data analysis/interpretation, and reporting.
Contact Us
Please contact the Core at the conceptual stage of research to ensure quality results.
To request biostatistical support for your research, please contact Senior Biostatistician, Dr. Beverly J. Spray, PhD, by email or phone (501-364-3184).
The purpose of the Arkansas Children's Research Informatics Core (RIC) is to support investigators and researchers to gain access to state-of-the-art research tools and technologies for biomedical research.
About Us
The Research Informatics team at Arkansas Children’s Research Institute offers the services and the tools that enable investigators to conduct research activities. The Research Informatics team emphasizes biomedical informatics as an approach in facilitating research projects and in assisting investigators in the daily use of research database systems and clinical/operational support requests. The Research Informatics team maintains state-of-the-art research tools and technologies.
Our Services
Data Management
- Manage the life cycle of research informatics projects:
- Define research and project needs with investigators and manage the delivery of project deliverables.
- Devise and implement policies and standard operating procedures with attention to data security and efficiency of data handling and management.
- Manage research informatics tools such as RAiA, TriNetX, and ACT as well as assist in the development of the Research Data Capture (REDCap) and PHIS systems.
Development
- Retrieve data for approved research, quality improvement, and operational support projects, primarily through the electronic medical record repository as well as other resources such as RAiA, TriNetX, and the Children’s Hospital Association’s (CHA) Pediatric Health Information System (PHIS), among others.
- Define ways to organize, store, and analyze data with attention to security and confidentiality.
Support and Training
- Support Arkansas Children’s and UAMS investigators in daily use of research database systems.
- Develop and conduct training for Arkansas Children’s and UAMS investigators with a focus on teaching best practices to enhance data efficiency, collection, and security.
- Provide continuing service request support of database projects, including access, security, maintenance, and enhancement.
Core Staff
Request Our Services
Use the links below to initiate a request for services. A member of the Research Informatics team will contact you shortly after the request is submitted. If you have questions or need help, you can attend the Ask RIC sessions held every Monday from 12 pm to 1 pm.
We look forward to providing you with cutting-edge informatics services.
Contact Us
To request informatics support for your research, please contact the research informatics program manager, Erin C. Hathorn, MSHI.

ACT Network
ACH is part of the Accrual for Clinical Trials (ACT) Network, a nationwide group of leading academic research institutions that share patient counts from electronic health record data. Explore and validate feasibility for clinical studies to learn more about patient populations.
Learn moreACRI and UAMS have established the Division of Child Health Biomedical Informatics within the UAMS Department of Biomedical Informatics. Housed at ACRI, Child Health Biomedical Informatics provides a focal point for team science research involving state-of-the-art quantitative analytics across the spectrum from molecules (bioinformatics) to human populations (public health informatics). This team is an academic focal point for informatics professionals with primary appointments in other departments and institutes. Division faculty also work closely with ACH Information Technology to define network, storage, and computational capabilities to support the ACRI research community. Current projects include establishing ACRI high-performance computing, private cloud computing, petabyte-scale storage systems, and a research data repository. In collaboration with ACH Clinical Informatics, Child Health Biomedical Informatics is also working to establish the ARLife resource that enables researchers to track parent/child dyads across the ACH and UAMS electronic health records and the Arkansas All Payers Claims Database. The Child Health Biomedical Informatics collaborates with the ACRI Biostatistics Core to provide research design consultation, data management, and machine learning-based analyses.
At ACRI, maintaining the highest standards of biosafety is essential to ensuring a safe and compliant research environment for all staff, trainees, and collaborators. Our Research Biosafety Program provides guidance, oversight, and support for research involving biological materials, including recombinant or synthetic nucleic acids, infectious agents, and other biohazardous materials. We are committed to promoting a culture of safety, regulatory compliance, and responsible research practices.
If you need additional information or have questions regarding Research Biosafety at ACRI, please contact the Research Biosafety Specialist and the Research Biosafety Committee, ResearchBioSafetyCommittee@archildrens.org.
As part of our commitment to transparency and compliance with federal guidelines, ACRI has implemented procedures to ensure public access to Institutional Biosafety Committee (IBC) meeting minutes. In accordance with recent NIH guidance, external IBC administered for ACRI will publicly post approved minutes for IBC meetings occurring on or after June 1, 2025. These minutes will be made available immediately following approval and after all appropriate and allowable redactions have been completed.
ACRI utilizes external administered Institutional Biosafety Committee (IBC), please use the links below to access IBC meeting minutes as they pertain to ACRI:
- The minutes for the external IBC administered by UAMS IBC are located here: https://uams.edu/campusopsstaging/depts/ohs/biosafety_cmte
- The minutes for the external IBC administered by Advarra minutes are located here: https://ibcminutes.com/AR00036
Additional Resources
-
Resource
Shared Equipment and Location
Bookmark Shared Equipment and LocationListing of Shared Equipment and Location -
Research
Information Systems
Bookmark Information SystemsA list of information systems to support ACRI research. -
Research
ACRI Institutional Information
Bookmark ACRI Institutional InformationInstitutional information is available for use in completing grant application forms. -
Research
Intramural Grant Programs
Bookmark Intramural Grant ProgramsView intramural Grant Opportunities at ACRI. -
-
Research
Pediatric Clinical Research Unit (PCRU)
Bookmark Pediatric Clinical Research Unit (PCRU)Learn About the PCRU at Arkansas Children's
-
Research
Using Human Specimens, Cell Lines or Data
Bookmark Using Human Specimens, Cell Lines or DataFAQ about Research Using Human Specimens, Cell Lines or Data -
Research
COI Disclosure, Education, and Policies
Bookmark COI Disclosure, Education, and PoliciesConflict of Interest Disclosure, Education, and Corporate Compliance Policies -
Research
Using Controlled Substances in Research
Bookmark Using Controlled Substances in ResearchGet guidance and information about using controlled substances in research.
-
Research
NICHD Data and Specimen Hub (DASH)
Bookmark NICHD Data and Specimen Hub (DASH)DASH is a centralized resource for researchers to share and access de-identified data from NICHD-funded studies.
-
Research
ACRI Resource List
Bookmark ACRI Resource ListFind the resources you need! Click the link below to choose a category or search by keyword.
-
Research
Starting a Research Project?
Bookmark Starting a Research Project?Begin your research project with ease using our intake/feasibility form. Note: UAMS or Arkansas Children's email address required.

